2019-nCoV and B.1.617.2 Variant Nucleic Acid Test Kit (PCR- fluorescence probe method)
Advantages
Sample: Human nasopharyngeal swabs, oropharyngeal swabs, sputum, alveolar lavage fluid specimens.
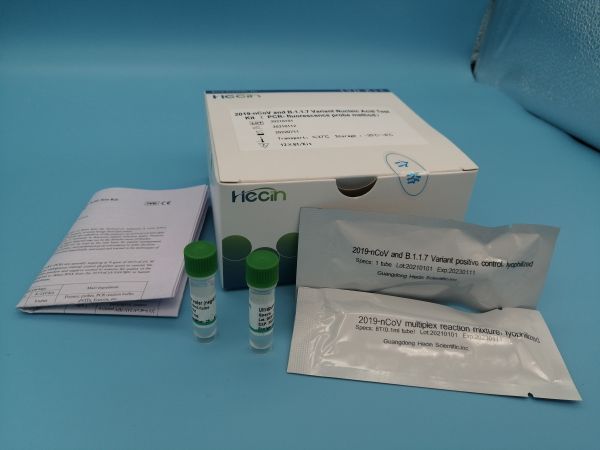
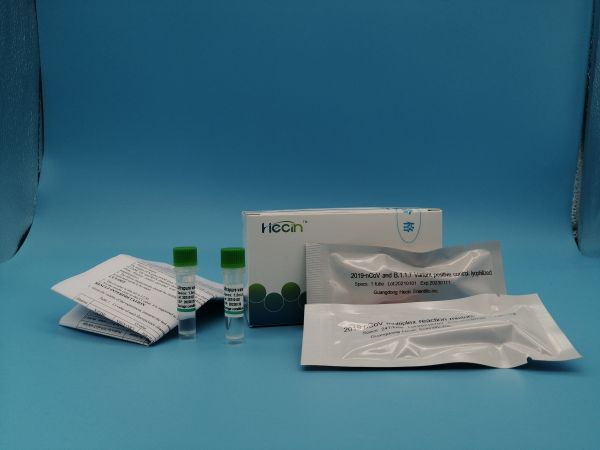
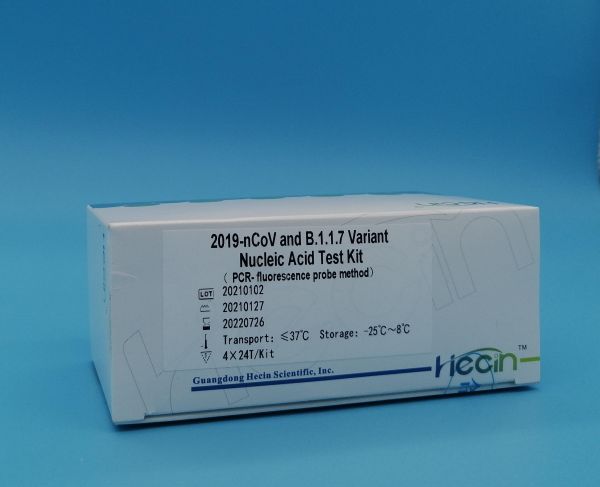
Specification:12×8T/Kit;4×24T/Kit;
Storage conditions and validity period: Unopened Kit: Store between -25℃ to 8℃ away from light, valid for 18 months.
Dye:FAM (for N gene), VIC (for T478K), ROX(L452R),CY5 (for internal standard)
Features
N and T478K, L452R target: the three targets improve the positive detection rate, greatly reduce the rate of missed detection.
The positive control is a pseudovirus particle: Simulating the novel coronavirus structure and playing a real control role.
Minimum detection limits: 400 copies/mL for 2019-NCOV, 400 copies/mL for 2019-NCOV T478K mutation and 1000 copies/mL for 2019-NCOV L452R mutation.
Negative and positive controls were set up, and an internal standard system was introduced, which can greatly reduce the false positives caused by operation
Advantages of freeze-dried powder: easy to transport, stable reagent performance, no repeated freeze-thaw problems.
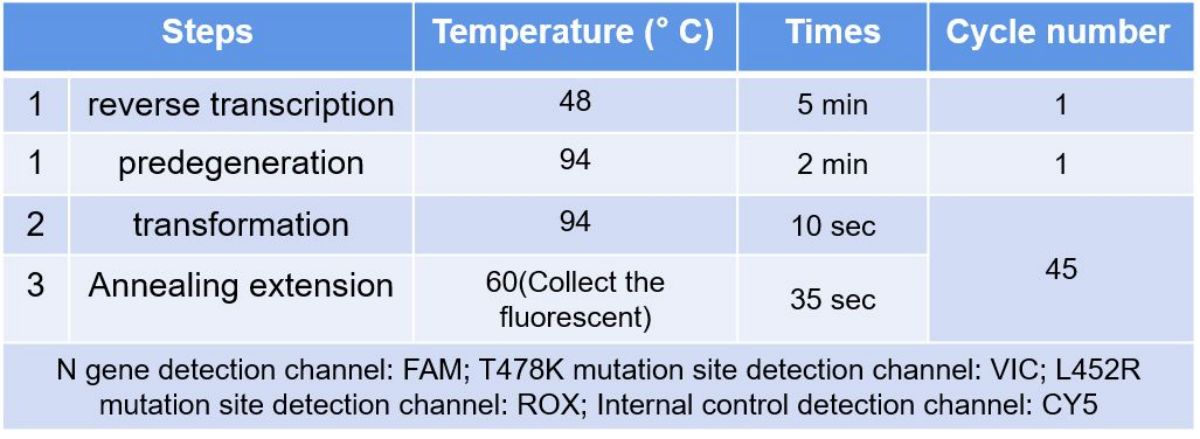
Instrument setting
Reaction tubes/plates were placed in the instrument and four channels were selected for FAM, VIC, ROX and CY5 per sample. The specific program Settings are as follows:



Product specification
8X12
24X2
HS33- 12X8T/Kit
HS33- 4X24T/Kit