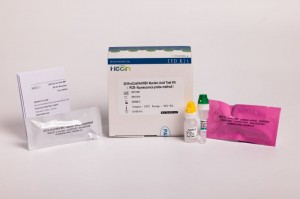
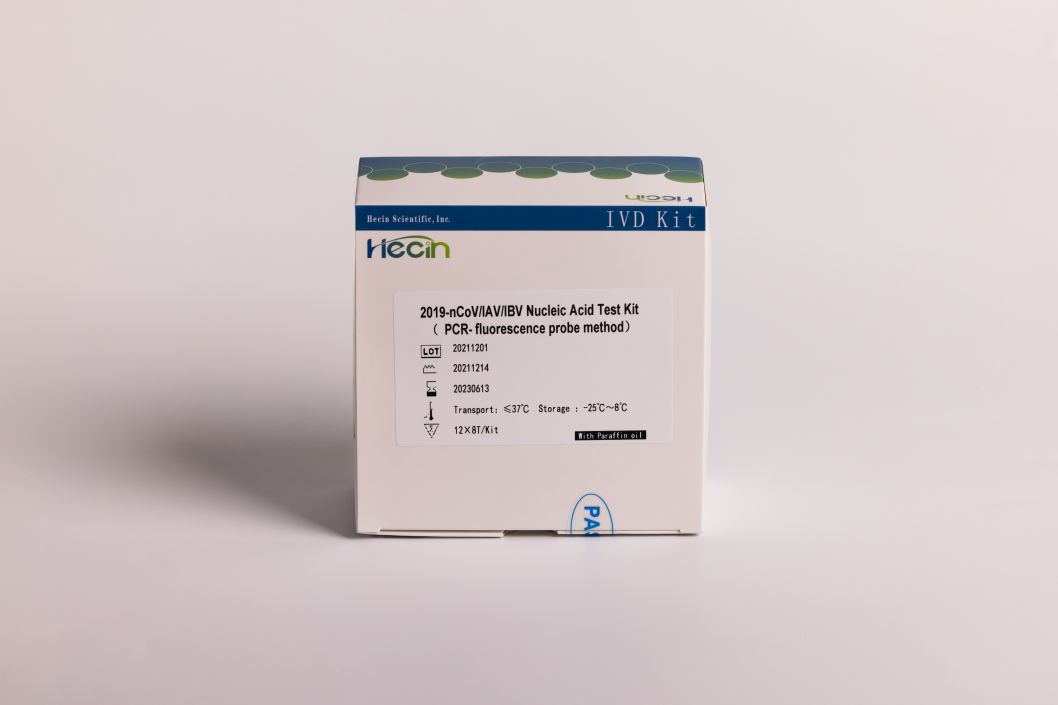
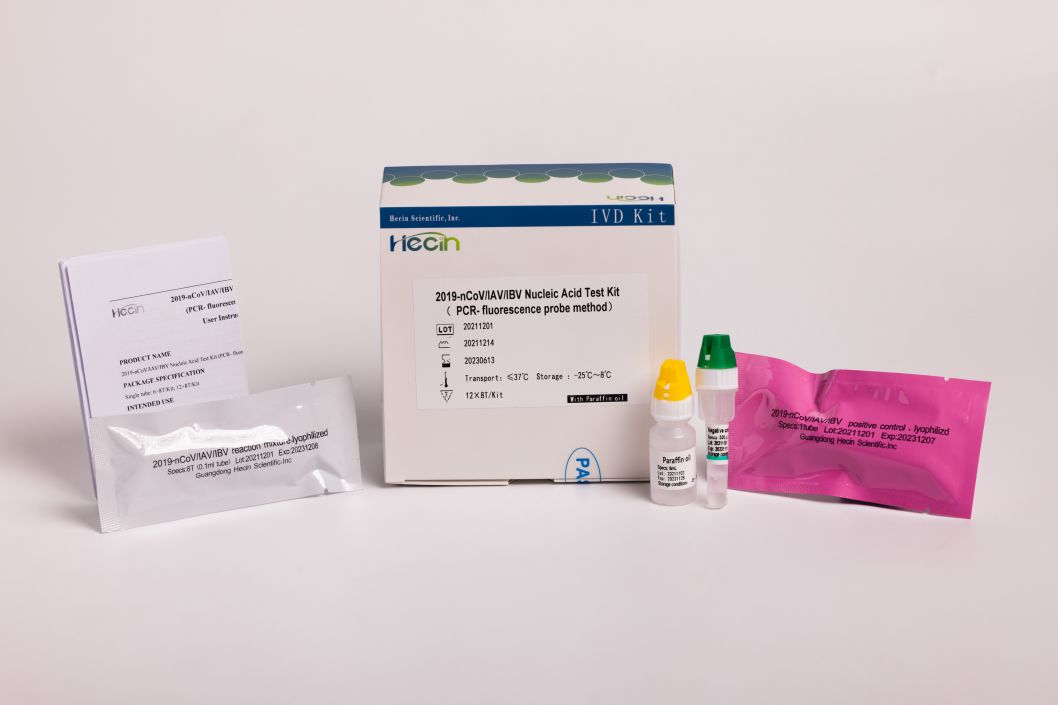
2019-nCoV/IAV/IBV Nucleic Acid Test Kit (PCR- fluorescence probe method)
Intended Use
This kit is a real-time RT-PCR test intended for the qualitative detection of RNA from the 2019-nCoV, Influenza A virus (IAV), Influenza B virus (IBV).
Advantages
Cooperative detection: qualitative detection of RNA from the 2019-nCoV, Influenza A virus (IAV), Influenza B virus (IBV).
Transportation within 37 ℃ for 3 months: lyophilized reagent is more stable;Transportation conditions: ≤37℃, stable for 3 months.
Less operation: The amplification reaction solution was premixed together, reduce laboratory operations.
Different types of sample: Specimen type nasopharyngeal swab; oropharyngeal swabs;sputum;alveolar lavage fluid specimens.
Reliable Performance: Coincidence rate of positive or negative reference products: 100%. the coefficient of variation of Ct value (CV, %) is less than or equal to 5%.
Instruments
UltraFast QPCR FQ-8A,QuantGene 9600 Real-Time PCR System ;ABI 7500 Real-Time PCR System, Roche LightCycler96 Real-Time PCR System, Tianlong TL988 Real-Time PCR System, SLAN Real-Time PCR System etc.
Fluorescent channel required
N gene detection channel: FAM
IBV detection channel: VIC
IAV detection channel: Tex Red
Internal control detection channel: CY5
Limit of Detection
2019-nCoV : 400 copies/mL.
Influenza A virus : 1.5 TCID50/mL.
Influenza B virus : 2.0 TCID50/mL.
Analytical specificity
This test kit can be used to detect 2019-nCoV, Influenza A virus (2009 H1N1,H1N1,H3N2,H5N1, H7N9), Influenza B virus (Yamagata, Victoria) specifically.



Product specification
12X8T
12X4T
HS28- 12X8T/Kit
HS28- 4X24T/Kit